Your Location:Home > Products > Organic Intermediates > pymetrozine


CasNo: 123312-89-0
MF: C10H11N5O
Appearance: colorless crystals
|
Descriptioin |
Pymetrozine is a new neuroactive insecticide. It has a remarkable selectivity for sucking insect pests, such as aphids, whiteflies, and plant hoppers, due to its systemic action. Pymetrozine is the only representative of the pyridine azomethines, a new class of insecticides. Pymetrozine has been determined to be of low acute toxicity to humans, birds, aquatic organisms, mammals, and bees. It is also not mutagenic. |
|
Uses |
Pymetrozine is a systemic insecticide used in agriculture to control many pests, including aphids, whiteflies, and other sucking insects: Aphids: Pymetrozine is effective against many strains of aphids on potatoes, leafy vegetables, and other brassica crops Whiteflies: Pymetrozine can reduce whiteflies on ornamental plants in greenhouses Other sucking insects: Pymetrozine is effective against all life stages of sucking insects, including planthoppers and leafhoppers |
|
Definition |
ChEBI: A member of the class of 1,2,4-triazines that is 4,5-dihydro-1,2,4-triazin-3(2H)-one substituted by a methyl group at position 6 and a (pyridin-3-ylmethylidene)amino group at position 4. |
InChI:InChI=1/C10H11N5O/c1-8-7-15(10(16)14-13-8)12-6-9-3-2-4-11-5-9/h2-6H,7H2,1H3,(H,14,16)/b12-6+
A practical four-step synthesis of pymet...
The conclusions were reached on the basis of the evaluation of the representative uses of pymetrozine as an insecticide on tomato, aubergine, potato and oilseed rape. The reliable endpoints concluded as being appropriate for use in regulatory risk assessment, derived from the available studies and literature in the dossier peer reviewed, are presented. Missing information identified as being required by the regulatory framework is listed. Concerns are identified.
Pymetrozine is a neuroactive insecticide but its site of action in the nervous system is unknown. Based on previous studies of symptoms in the locust, the feedback loop controlling the femur–tibia joint of the middle leg was chosen to examine possible targets of the insecticide.
The crystals of functionalized pymetrozi...
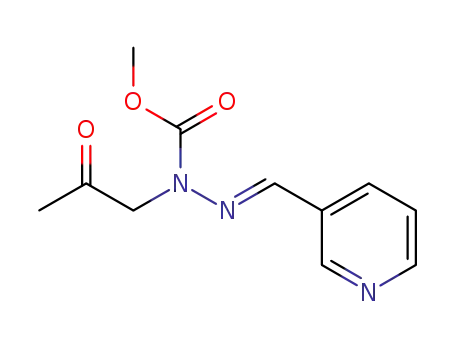
methyl (E)-1- (2-oxopropyl)-2-(pyridin-3-ylmethylene)hydrazine-1-carboxylate

C11H15N5O2

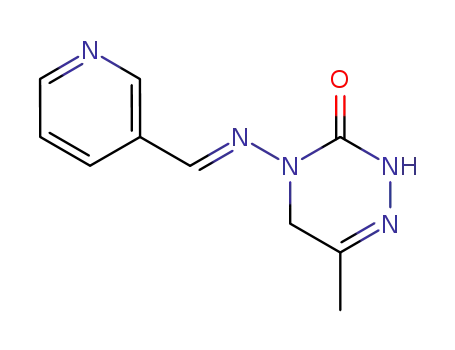
pymetrozine

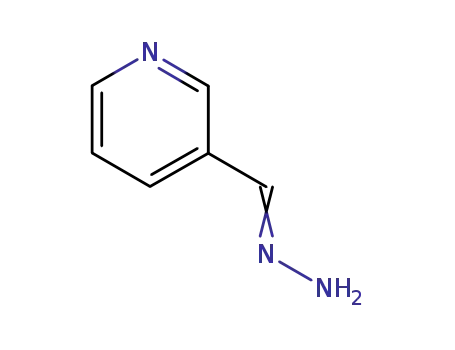
hydrazone of pyridine-3-carboxaldehyde

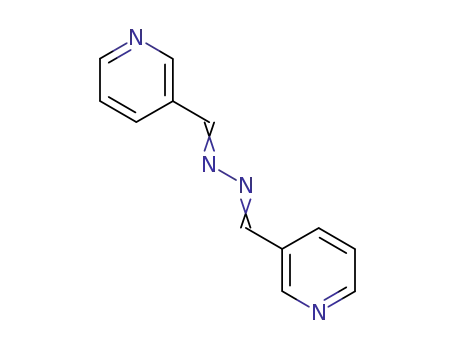
1,4-bis(3-pyridyl)-2,3-diaza-1,3-butadiene
| Conditions | Yield |
|---|---|
|
With hydrazine hydrate; In ethanol; at 25 ℃;
|
37% 28% 15% 14% |
|
With hydrazine hydrate; In ethanol; at 80 ℃; Solvent;
|
16% 36% 18% 21% |
|
With hydrazine hydrate; In ethanol; at 80 ℃;
|
15% 35% 19% 17% |
|
With hydrazine hydrate; In ethanol; at 50 ℃;
|
21% 24% 10% 8% |
C10H12ClN5O


pymetrozine
| Conditions | Yield |
|---|---|
|
With potassium carbonate; In methanol; at 25 - 30 ℃; for 5h; Reagent/catalyst;
|
85% |
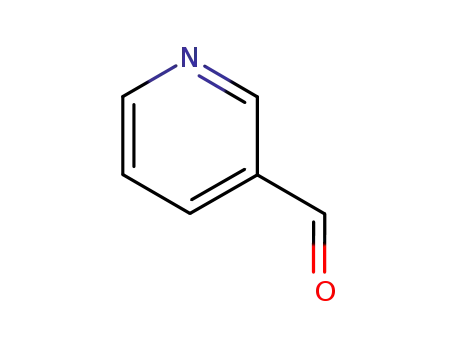
3-pyridinecarboxaldehyde
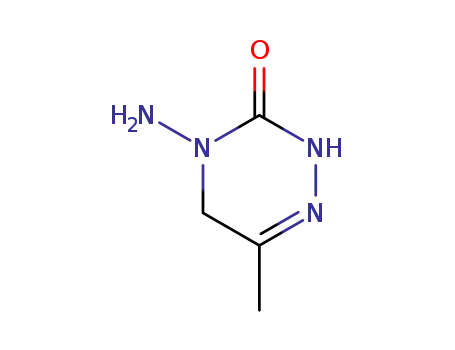
2,3,4,5-tetrahydro-3-oxo-4-amino-6-methyl-1,2,4-triazine
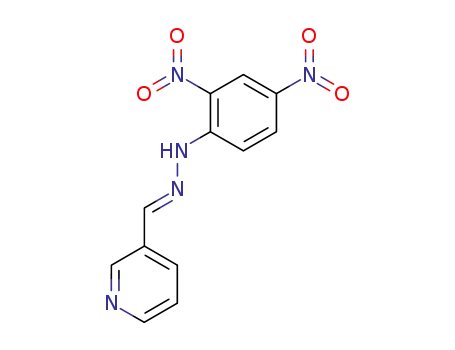
pyridine-3-carbaldehyde-(2,4-dinitro-phenylhydrazone)

2,3,4,5-tetrahydro-3-oxo-4-amino-6-methyl-1,2,4-triazine